Questions on synthetic strategies to transfer self-assembly in multi-step noncovalent synthesis are still in their exciting early stages. Seeking the highest possible control for the construction of functional adaptive systems with well-defined morphologies, a supramolecular chemist must come up with new methodologies to perform noncovalent synthesis (see Science 2019, 363, 1396-1397), replacing the current stochastic method of exploration. In order to do so, the understanding of the how-and-what molecules influence to assemble, is of unbearable importance. To tackle these fundamental questions, we are ever since interested into designing and studying new building blocks, understanding their mechanism of assembly, amplification of asymmetry, the role of external factors such as temperature, solvents and humidity and more recently, the combination of covalent and non-covalent reactions in supramolecular polymers. Next to many advanced experimental techniques, we strengthen our conclusions with computation studies to simulate the experiments. With every system we study, we thrive to add little pieces to the toolbox, to ultimately be able to control supramolecular processes and to truly call ourselves supramolecular chemists. To initiate this paradigm shift is to recognize that the sample preparation has a huge impact on the structures formed, and it requires rigorous reporting of the details in publishing. In this section we have four closely related lines of research replacing the current stochastic method of exploration of available structure-energy combinations of the system. In order to tackle this fundamental question, we are focusing on supramolecular polymers (see the section Supramolecular polymers; we’ve come full circle).
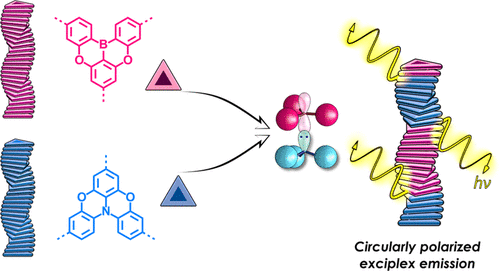
Mechanisms of cooperative self-assembly processes
By studying the one- and two-component polymerization of disc-like molecules experimentally as well as theoretically, we have quantified the thermodynamic characteristics of a wide range of polymers. An important goal is the ability to predict how the molecular structure translates to the nature of the self-assembly process and stability of the aggregates formed, leading to new motifs and supramolecular polymers that can be used in the other two topics of the group. Moreover, we like to arrive on a full understanding of the role of solvent and other external factors such as temperature and humidity. In many cases we use chirality to study the cooperative nature of the supramolecular polymerization. Analytical methods such as circular dichroism spectroscopy as well as UV/Vis spectroscopy turn out to be excellent techniques to get insights into the details of supramolecular polymerizations and the arrangement of the monomers in the polymer.
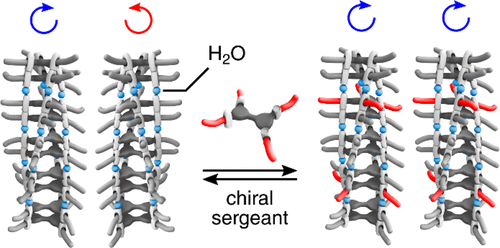
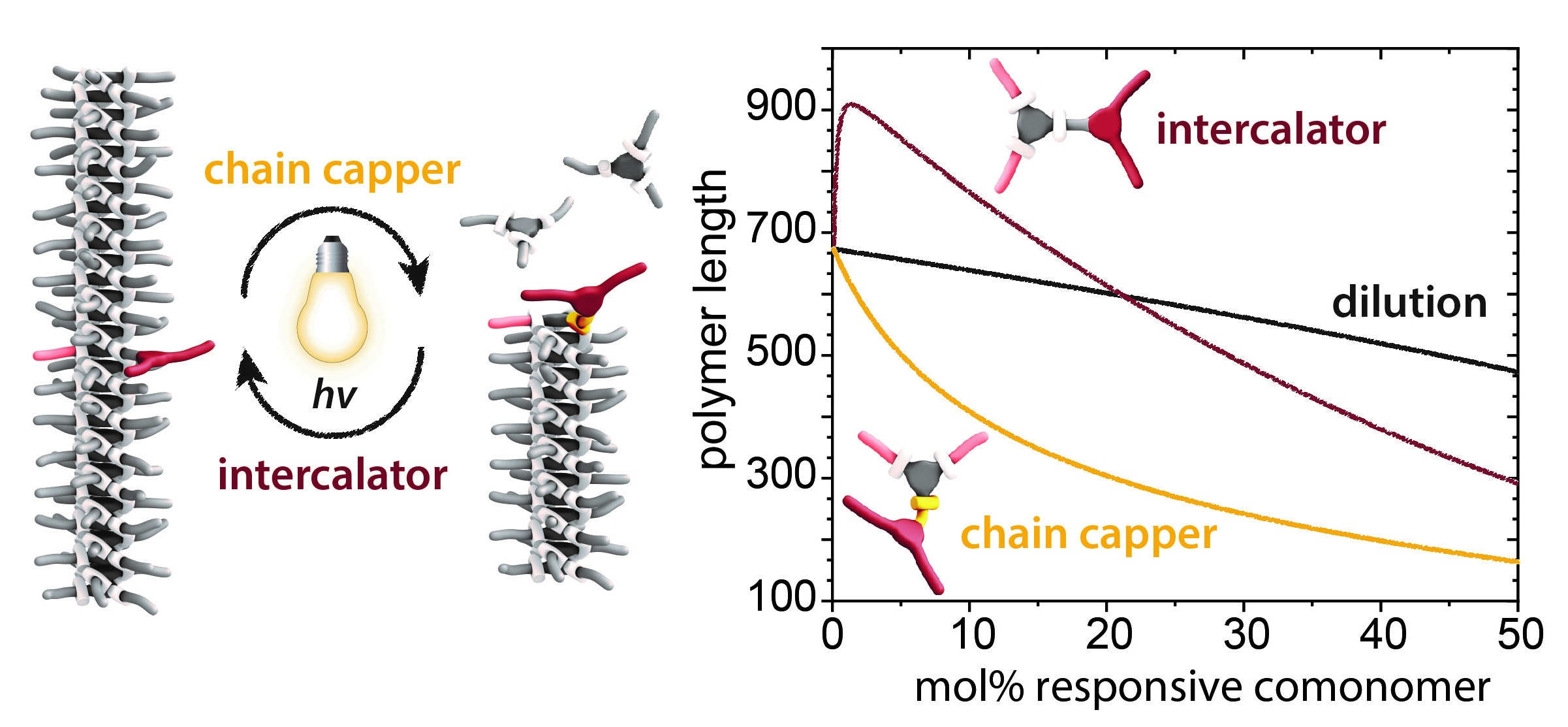
Controlling the length and stereochemistry of supramolecular polymers
Our ambition is to control the microstructure in solution, followed by studying how the microstructure will behave in bulk materials. A combination of experimental techniques and mathematical models permits to elucidate the thermodynamics and kinetics of polymerization processes of one, two, or more monomers. While homo polymerizations are important to study the fundamentals of, per example, a new core, copolymerizations bear many exciting possibilities to further modify the formed microstructures. Comonomers added to a supramolecular polymer can act as simple comonomer, but more interestingly as intercalator, chain-stopper or sequestrator; they have all another role in tuning length, sequence and stereochemistry of supramolecular polymers.
Combining covalent and non-covalent synthesis of molecular systems
Like in covalent synthesis, the outcome of a non-covalent synthesis reaction is determined by the order of events. We currently design multiple component systems, that have various types of interactions to arrive at adaptive functional supramolecular materials. An important novel concept in the group is the combination of covalent and non-covalent synthesis. Here we like to control the assembly as the result of a covalent modification, as well as stabilizing a supramolecular aggregate by a chemical reaction. Preliminary studies are aiming at mimicking the synthesis of the collagen triplet helical bundles, which are only formed after modifying the soluble precursors. These studies will lead to a bottom-up approach for synthesizing stable, highly functional systems and materials. This very fundamental investigations are crucial aspects of understanding some of the essential steps in the origin of life.
Novel concepts
Next to fundamentals of supramolecular polymers and systems, we are always fascinated by the unexpected and out-of-the-box concepts. As a result, we continuously design, synthesize and characterize novel structures and study them in solution and on surfaces. Recent examples can be found in our studies to explore boron-nitrogen interactions and the use of nitrogen-rich discotic molecules with cleavable solubilizing tails.